Your health journey is unique — and so is our approach to women’s care. From adolescence through menopause and beyond, our team provides expert support tailored to your body, goals and lifestyle.
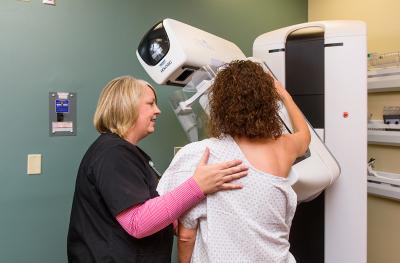
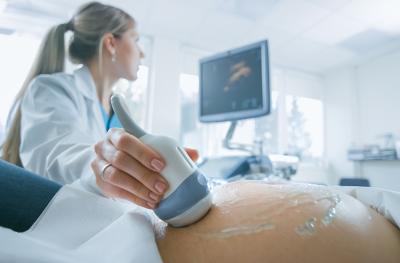
Whether you're planning a pregnancy, managing a complex condition or simply keeping up with routine screenings, we’re here to guide you every step of the way. Our women’s health specialists offer a full range of services, including fertility and pregnancy care, gynecologic wellness, breast health and menopause support — all designed with your comfort and preferences in mind.