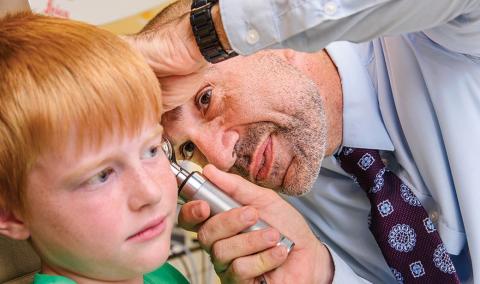
When your child is experiencing health problems related to their ears, nose, throat, head or neck, it can affect their entire life. Children’s Hospital offers the only pediatric ENT (ear, nose and throat) clinic in mid-Missouri.

Our pediatric experts have specialized training in the care of kids. They are focused exclusively on the diagnosis and treatment of children with conditions affecting their breathing, swallowing and feeding, sleep, and ears and hearing.
Pediatric ENT conditions we treat
Noisy breathing
Depending on your child's age, noisy breathing can be caused by issues in the voice box (larynx), breathing tube (trachea), or lungs.
Stertor, stridor and wheezing. These terms refer to different sounds that are often described as "noisy breathing" depending on where in the respiratory tract the problem is occurring.
- Stertor is a noise from the back of the nose or throat that sounds like nasal congestion.
- Stridor is a high pitch, sometimes "sqeaky" sound, and often is caused by issues with or near the voice box. It can be heard when your child breaths in or out — or both.
- Stridor can be a sign of subglottic stenosis, which involves narrowing of the airway just below the vocal cords, or tracheal stenosis, narrowing of the windpipe (trachea). These conditions can be present at birth (genetic) or can develop in response to infection, irritation, or injury (acquired).
- Stridor might also be a symptom of vocal cord dysfunction or paralysis. This might be treated with voice therapy, injections, surgery or a mix of the three. If vocal cord dysfunction is present, this stridor is often heard during exercise.
- Wheezing is a persistent sound that often is related to problems in the lungs. It usually only happens when your child is breathing out.
Laryngomalacia, tracheomalacia and bronchomalacia. These conditions refer to abnormalities with the voice box (laryngomalacia), trachea (tracheomalacia) and lungs (bronchomalacia) that can cause noisy breathing. Symptoms can include irritability, difficulty breathing, poor eating and poor weight gain (or weight loss).
Laryngomalacia, is the most common cause of noisy breathing in infants. It generally improves as the infant ages and the airway grows and becomes stronger. However, if the child has severe breathing difficulties or trouble eating and gaining weight, this condition might require medication or surgery.
Our pediatric ENT experts offer many treatment options for noisy breathing. Some conditions only need to be monitored and might resolve with time. Others might require medication or surgery. Our pediatric experts have the knowledge needed to evaluate the cause of noisy breathing and will discuss all possible treatment options with you and make a recommendation for the best treatment for your child.
Swallowing and feeding issues
When children have issues swallowing, known as dysphasia, it often affects their ability to eat enough, and that can lead to irritability, pneumonia, failure to gain weight (or weight loss) or, in rare cases, hospitalization.
Dysphagia can happen in one of the three phases of the feeding process:
- An infant might have trouble sucking on the bottle or latching onto the breast, while an older child might have trouble chewing.
- One common reason a baby can't latch to breastfeed is tongue-tie (ankyloglossia), which reduces the tongue's range of motion. This can be fixed through surgery, if needed.
- When swallowing, the child must close off their airway for protection. If there is a problem doing this, the child might repeatedly choke or cough while trying to swallow.
- Once the food makes it to the food pipe (esophagus), it should then move on to the stomach. When this doesn't happen, the child might spit up or throw up. This condition is known as gastroesophageal reflux (or GERD). This is a problem when the material that is thrown up also enters the airway.
Signs of dysphagia include repeated coughing or choking while feeding, frequent upper respiratory illnesses, wheezing, wet breath sounds, spitting up during meals or throwing up after meals, and liquid coming out of the nose or mouth during feeding. Our team offers a variety of treatment options depending on the type and severity of the dysphagia.
Laryngeal cleft, laryngotracheal cleft, laryngotrachealesophageal cleft. These are a group of rare inherited disorders in which there is an abnormal opening between the airway and the esophagus. This can cause children to inhale food into their airway. These conditions require surgery to repair, and this can significantly improve a child's swallowing function.
Other swallowing and feeding conditions we treat
- Acid reflux
Sleep problems
Good sleep is important to a child's growth and development. Our pediatric ENT doctors work alongside the Children's Hospital pediatric sleep medicine team to help diagnose and treat your child's sleep problem.
If your child has large tonsils or adenoids that are causing snoring, mouth breathing, or gasping for breath during sleep, they may have a condition called obstructive sleep apnea. In children, a tonsillectomy and/or adenoidectomy can improve these symptoms to help your child breath and sleep easier. Our pediatric ENT doctors can evaluate your child and determine if this surgery would be the right treatment for your child or if your child would benefit from additional evaluation.
Learn more about pediatric sleep medicine.
Ear and hearing issues
Seventy percent of all children in the United States will have a middle ear infection (acute otitis media) by the time they are 2 years old. This condition affects the air-filled space behind the eardrum, resulting in ear pain, fever, and possibly fluid drainage from the ear.
Ear infections are caused by an infection, either bacteria or virus, that often accompanies another illness such as a cold, flu, or allergy. The infection causes congestion and swelling of the nasal passages and throat as well as the eustachian tubes. These tubes run from the middle ear to the back of the throat and regulate air pressure and drain fluid from the middle ear.
Ear infections often resolve on their own, or your doctor might prescribe antibiotics if the infection is bacterial. Because children's eustachian tubes are narrow and often get clogged, some children experience frequent ear infections related to lack of airflow and drainage in the middle ear.
In this case, our pediatric ENT doctors can perform ear tube surgery to implant tubes (called myringotomy tubes, tympanostomy tubes, or pressure equalization tubes) in your child's eardrum that will allow proper drainage and airflow. If you think your child might need ear tube surgery, our doctors can help you determine whether that is the right treatment.
Other ear conditions we treat
- Balance disorders or vertigo
- Bone growth in ear (otosclerosis)
- Cysts in the ear (cholesteatoma)
- Ear drainage (otorrhea)
- Hearing loss
- Ringing in the ears (tinnitus)
- Ruptured eardrum (tympanic membrane perforation)
- Swimmer's ear (otitis externa)
Other conditions we treat
- Drooling and bad breath
- Facial and neck trauma (bites, lacerations, fractures)
- Head and neck masses
- Hemangioma and vascular malformations
- Nasal disorders
- Sinus infection (sinusitis)
- Tonsil infection (tonsillitis) or recurrent strep pharyngitis
- Trachea infection (tracheitis)
- Voice and speech disorders
Mid-Missouri's most experienced pediatric ENT experts
Kids are not just little adults. Their bodies are different, and so the conditions that affect their ears, nose and throat must be treated differently, too. Our doctors have experience treating the simplest to the most complex ear, nose, and throat problems in children, and they are committed to the overall health and well-being of your child.
As part of Children's Hospital, our doctors work closely with other children's health experts to provide comprehensive care tailored to the needs of your child. They will get to know you and your child to determine the cause of your child’s problem, and they will offer the best treatment to make them feel better sooner.
No matter your child's treatment, our team will be by your side throughout the process, providing follow-up visits to ensure your child is doing well.
Related Conditions & Treatments
- Adolescent Medicine
- Aerodigestive Program
- Down Syndrome
- Emergency Care for Kids
- Feeding Tubes for Children
- Juvenile Diabetes
- Hyperbaric Oxygen Therapy
- Inpatient Pediatric Psychiatric Care
- Neonatology
- Pediatric Anesthesiology
- Pediatric Cancer
- Pediatric Cardiology
- Pectus Carinatum
- Pediatric Chest Wall Disorders Program
- Pediatric Dermatology
- Pediatric Development and Behavior
- Pediatric ENT (Ear, Nose and Throat)
- Pediatric Epilepsy
- Pectus Excavatum
- Pediatric Eye Care
- Pediatric Gastroenterology
- Pediatric Infectious Diseases
- Pediatric Inpatient Rehabilitation
- Pediatric Nephrology
- Pediatric Neurology
- Pediatric Neurosurgery
- Pediatric Orthopaedics
- Pediatric Plastic Surgery
- Pediatric Primary Care
- Pediatric Psychiatry
- Pediatric Pulmonary Medicine
- Pediatric Sleep Medicine
- Pediatric Surgery Services
- Pediatric Surgical Services
- Pediatric Urology
- Pediatric Vascular Anomalies
- Pediatric Weight Management
- Sickle Cell Disease